Ion channels are pore-forming protein complexes that facilitate the flow of ions across the hydrophobic core of cell membranes. They are present in the plasma membrane and membranes of intracellular organelles of all cells, performing essential physiological functions including establishing and shaping the electrical signals which underlie muscle contraction/relaxation and neuronal signal transmission, neurotransmitter release, cognition, hormone secretion, sensory transduction and maintaining electrolyte balance and blood pressure. They are usually classified by gating i.e. the stimulus that ‘opens’ the channel, be it chemical or mechanical stimuli.
Most Na+, K+, Ca2+ and some Cl– channels are gated by voltage, whereas others (such as some K+ and Cl– channels, TRP channels, ryanodine receptors and IP3 receptors) are relatively voltage-insensitive and are gated by second messengers and other intracellular and/or extracellular mediators.
Many ion channels (e.g. K+, Na+, Ca2+, HCN and TRP channels) share several structural similarities which suggests that they have evolved from a common ancestor. This group of ion channels can be classified together as the ‘voltage-gated-like (VGL) ion channel chanome’ (Figure 1).
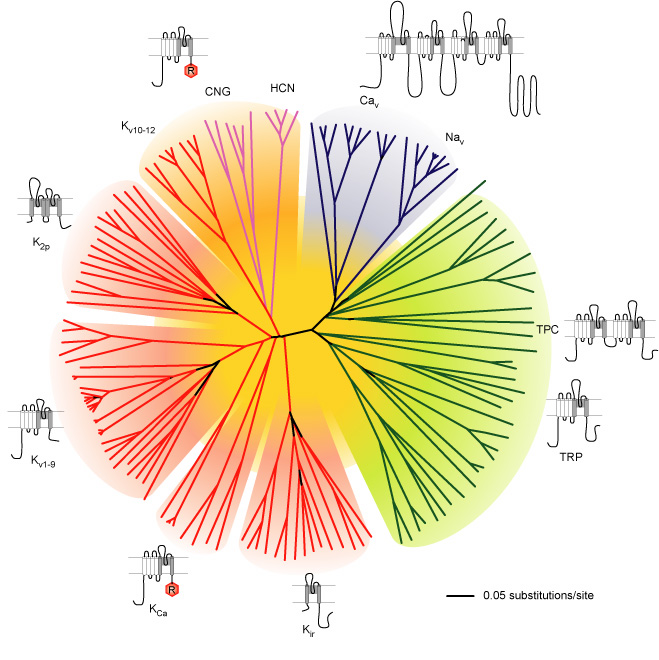
Figure 1. Representation of the amino acid sequence relations of the minimal pore regions of the voltage-gated ion channel superfamily. This global view of the 143 members of the structurally related ion channel genes highlights seven groups of ion channel families and their membrane topologies. Four-domain channels (CaV and NaV) are shown as blue branches, potassium selective channels are shown as red branches, cyclic nucleotide–gated channels are shown as magenta branches, and transient receptor potential (TRP) and related channels are shown as green branches. Background colors separate the ion channel proteins into related groups: light blue, CaV and NaV ; light green, TRP channels; light red, potassium channels, except KV10–12, which have a cyclic nucleotide–binding domain and are more closely related to CNG and HCN channels; light orange, KV10–12 channels and cyclic nucleotide–modulated CNG and HCN channels (from Yu and Catterall, 2004)
Other ion channels, such as Cl channels, aquaporins and connexins, have evolved separately and possess completely different structural properties to the VGL channels.
Defects in ion channel function cause a wide range of disorders termed ‘channelopathies’ which include conditions resulting from mutations in ion channels (e.g. cystic fibrosis, long QT syndrome, short QT syndrome- inherited ion channel diseases are reviewed by Lieve and Wilde (2015)) and acquired diseases caused by autoimmune attack on ion channels (e.g. myasthenia gravis and possibly multiple sclerosis- autoimmunity and channelopathy are reviewed by RamaKrishnan and Sankaranarayanan (2016)).
Ion channel modulators are an extremely successful drug class, second only to drugs targeting G protein-coupled receptors, with amlodipine, zolpidem, alprazolam, the sulfonylureas, repaglinide and nateglinide amassing huge returns for their developers. Technical advances in high-throughput screening methodology and high resolution crystal structures of ion channels should enable development of the ion channel drugs of the future. Potential new ion channel drug targets are discussed in Bagal et al. (2013).
Ligand-gated ion channels
Ligand-gated ion channels (LGICs) mediate passive ion flux driven by the electrochemical gradient for the permeant ions. LGICs are gated by the binding of a specific ligand to an orthosteric site(s) that triggers a conformational change that results in the conducting state, or by binding of endogenous, or exogenous, modulators to allosteric sites. LGICs are responsible for fast synaptic transmission in the nervous system and at the somatic neuromuscular junction.
This group contains the excitatory, cation-selective, nicotinic acetylcholine (nAch), 5-HT3, ionotropic glutamate (NMDA, AMPA and kainate receptors) and P2X receptors and the inhibitory, anion-selective, GABAA and glycine receptors as well as acid-sensing (proton-gated) ion channels (ASICs), epithelial sodium channels (ENaC), IP3 receptor and the zinc-activated channel (ZAC). LGICs are generally heteromultimers, with subunits encoded by multiple genes. Multimeric combinatorial diversity leads to the wide variety of receptors reported, with differing pharmacological and biophysical properties and varying patterns of expression within the nervous system and other tissues. The pharmaceutical industry is striving to use this heterogeneity to develop new therapeutic agents with improved discrimination between receptor isoforms and reduced off-target effects.
The epithelial sodium channels (ENaC) mediate sodium reabsorption principally in the aldosterone-sensitive distal part of the nephron and the collecting duct of the kidney, and also in the lung epithelia. In the kidney, these channels are involved blood pressure regulation and are associated with many cardiovascular diseases. The ‘potassium-sparing’ diuretics amiloride and triamterene are ENaC channel blockers.
Acid-sensing (proton-gated) ion channels (ASICs) are involved in fear conditioning, memory formation, and pain sensation
Aberrant LGIC function is associated with many diseases. For example, overactivation of NMDA glutamate receptors may play a part in causing neurotoxic damage in the development of neurodegenerative disorders.
Many prescription drugs exert their effects by modulating the activity of LGICs. Some of the more common drug groups are described below:
Volatile anesthetic drugs used for general anesthesia primarily block the activity of GABA-, glutamate- and glycine-gated LGICs, but different agents have different effects on each receptor, and may block other LGICs. For example, sevoflurane is thought to act as a positive allosteric modulator of the GABAA receptor, but also acts as an NMDA receptor antagonist, potentiates glycine receptor currents, as well as nACh and 5-HT3 receptor currents. Desflurane also acts as a positive allosteric modulator of the GABAA and glycine receptors and as a negative allosteric modulator of nACh receptors. Sevoflurane and desflurane have largely replaced isoflurane except in economically undeveloped areas, where their high cost precludes use. Nitrous oxide, used in surgery and dentistry for its anaesthetic and analgesic effects, directly modulates a broad range of ligand-gated ion channels, and this likely plays a major role in many of its effects. It moderately blocks NMDA and β2-subunit-containing nACh channels, weakly inhibits AMPA, kainate, GABAC, and 5-HT3 receptors, and slightly potentiates GABAA and glycine receptors (see Emmanouil and Quock (2007): Advances in understanding the actions of nitrous oxide).
Gamma-aminobutyric acid analogs (the ‘gabapentinoids’) such as gabapentin and its conjugated prodrug gabapentin enacarbil (both used to treat epilepsy, neuralgic/neuropathic pain) and pregabalin (used to treat generalised anxiety disorder (GAD), neuropathic pain, post herpetic neuralgia) were thought to act as GABAA receptor agonists and cause inhibitory action like GABA. However, more recent analysis reveals high-affinity gabapentin binding sites on neuronal membranes, subsequently demonstrated to represent the α2δ protein, an accessory component of L-type calcium channels, encoded by gene CACNA2D1 or CACNA2D2– see Rogawski and Bazil (2007): New molecular targets for antiepileptic drugs: alpha(2)delta, SV2A, and K(v)7/KCNQ/M potassium channels. But whatever the mechanism, the effect of GABA analogs is to inhibit release of monoamine neurotransmitters including norepinephrine, substance P, and glutamate.
Acamprosate (used alongside behavioural therapy to manage alcohol abstinence in alcohol-dependent patients) is another GABA-like drug which appears to stimulate GABAergic inhibitory neurotransmission and antagonise the effects of excitatory amino-acids, particularly glutamate (via antagonism of NMDA receptors).
NMDA receptor antagonists (or channel blockers) such as ketamine (an anesthetic), dextromethorphan (a widely used OTC cough suppressant), phencyclidine (PCP, withdrawn from use as an anesthetic pharmaceutical), and nitrous oxide act principally via inhibition of glutamate NMDA receptors. Dextromethorphan is metabolised to the NMDA antagonist dextrorphan by CYP2D6. Many drugs of this class are used recreationally because of their psychoactive and dissociative effects.
nAch receptor modulators such as nicotine itself (as nicotine replacement therapy) and the nAch receptor antagonist varenicline are used to aid smoking cessation.
Other ion channel
Other ion channel families include the aquaporins (which also includes aquaglyceroporins), a family of chloride channels which includes the cystic fibrosis transmembrane conductance regulator (CFTR), the connexins and pannexins and the sodium leak channel, non-selective.
Genetic defects in aquaporin genes have been associated with several human diseases including abnormalities of renal water regulation, nephrogenic diabetes insipidus, loss of vision, onset of brain edema and starvation. Agre and Kozono (2003) and Schrier (2007) discuss the role of aquaporins in human health and disease.
The CFTR potentiator drug ivacaftor and the CFTR correcting drug lumacaftor are approved to treat a subset of cystic fibrosis patients. Crofelemer, a medication used to treat HIV-AIDS related diarrhea, is a CFTR activator. It also activates another unrelated chloride channel, CaCC, a calcium-activated chloride channel.
The connexin and pannexin genes encode gap junction proteins in humans, Gap junctions are essential for many physiological processes including cardiac and smooth muscle contraction, regulation of neuronal excitability and epithelial electrolyte transport. Connexins and pannexins allow the passive diffusion of molecules of up to 1,000 Daltons which can include nutrients, metabolites and second messengers (such as IP3) as well as cations and anions. Mutations in connexin genes are associated with the occurrence of a number of pathologies, such as peripheral neuropathies, cardiovascular diseases and hereditary deafness.
The sodium leak channel, non-selective (provisionally assigned as NaVi2.1) is structurally related to the voltage-gated sodium channel family (Nav1.1 – Nav1.9), but is voltage-insensitive and displays distinctive ion selectivity and pharmacological properties. NaVi2.1 is is widely distributed within the central nervous system and is also expressed in the heart and pancreas, and may mediate the tetrodotoxin-resistant and voltage-insensitive Na+ leak current (IL-Na) observed in many types of neuron (see Lu et al. (2007) for further details). The calcium channel blocker verapamil also blocks NaVi2.1.
Voltage-gated ion channels
Voltage-gated ion channels (VGICs) are responsive to changes in the local electrical membrane potential, and are critical for the function of excitable cells, such as neurons and muscle cells. VGICs are ion-selective, with separate channels identified for each of the major physiological ions- Na+, K+, Ca2+, Cl–. Each type of channel is a multimeric complex of subunits encoded by a number of genes. Subunit combinations vary in different tissues, with each combination having distinctive voltage dependence and cellular localization. Some VGICs are highly localized, such as the CatSper Ca channels, whose expression is restricted only to the principal piece of the sperm tail.
There are many drugs whose mechanism of action involves perturbation of VGIC activity. Some of the main classes of drugs are discussed below:
Calcium channel blockers (CCBs) -see also the topic ‘Calcium channel blocking drugs’ in the Cardiovascular system section of the Drugs module
Calcium-channel blockers are smooth-muscle relaxors having a negative inotropic effect on the working myocardial cells of the atria and ventricles, with inhibition of Ca2+ entry blunting the ability of Ca2+ to serve as an intracellular messenger.
The dihydropyridine class of CCB drugs block activity of L-type calcium channels. Examples of this drug class are amlodipine, felodipine, isradipine, lacidipine, nicardipine, and nimodipine which are used in the treatment of hypertension. In comparison to phenylalkylamine class CCBs such as verapamil, the dihydropyridines are relatively vascular selective in their mechanism of action in lowering blood pressure.
Sodium channel blockers
Class III antiarrhythmics are primarily sodium channel blocking agents, and include the prescription medicines dronedarone and amiodarone hydrochloride.
Many local anaesthetic agents are also sodium channel blockers, and include lidocaine, bupivacaine, prilocaine, mepivacaine, tetracaine and ropivacaine. Mechanistically these drugs bind to an intracellular portion of voltage-gated sodium channels blocking sodium influx into nerve cells, which prevents depolarization. Without depolarization, no initiation or conduction of a pain signal can occur.
Some anticonvulsants (antiepileptic drugs or AEDs) work at least in part, by blocking sodium channels. By inhibiting sodium (and/or calcium) channel activity, AEDs act to reduce the release of excitatory glutamate which is elevated in epilepsy and may also reduce γ-aminobutyric acid (GABA) secretion.
Whilst not strictly ion channel inhibitors, proton-pump inhibitors also block ion transport across the membrane. In this case by irreversibly blocking the H+/K+ ATPase transporter activity of the proton pump on the surface of gastric parietal cells. This action produces a pronounced and long-lasting reduction of gastric acid production. PPIs are the most potent inhibitors of acid secretion available, and have largely superseded histamine H2 receptor antagonists which have similar effects, but a different mode of action. Prescription PPIs include omeprazole, esomeprazole, pantoprazole, lansoprazole, and rabeprazole.
The Transient Receptor Potential (TRP) superfamily of channels are found in sensory receptor cells that are involved in heat sensation, taste, smell, touch, and osmotic and volume regulation.
RESOURCES
This four minute narrated animation explains how ion channels function to generate the neuronal action potential.

This is a more in-depth narrated presentation (40 minutes long) explaining the action potential, discussing resting membrane potential, depolarization and repolarization.